
Viral mutation vs. antiviral drugs
Viruses are organism-like organisms that use DNA or RNA as genetic material, have no independent nutritional metabolic system, and need to be parasitized in the host for replication and survival. During the symbiosis of viruses with their hosts in nature, some viruses can escape the host's immune defenses and lead to host morbidity and mortality. Viruses have a very high chance of mutating their genetic material, which can help them to escape the escalating host immune system. According to the doctrine of viral evolution, the history of viral development far exceeds that of human evolution, about which very little is known. With the development of molecular cell biology, more than 7000 types of viruses have been discovered, among which more than 300 viruses can infect humans. Viral infections account for 3/4 of infectious diseases and pose a serious threat to human health. Since the 1960s, broad-spectrum antiviral drugs have been available, but due to the very rapid mutation of viruses and the subsequent development of viral resistance and side effects, there are no specific and effective drugs for the clinical treatment of viral infections.
Ridcicevir - the hope of the people?
With COVID-2019 raging at the beginning of 2020, the development of specific anti-neo-coronavirus drugs is imminent. in a paper published in the New England Journal on February 1, it was reported that the first patient with neo-coronavirus pneumonia in the United States improved after deterioration with intravenous compassionate dosing of ridecivir (Remdesivir/GS-5734) [1]. on February 6, the ridecivir The "double-blind clinical trial" was clinically evaluated in 761 patients enrolled in the Jinyintan Hospital, the City Pulmonary Hospital and the Union Hospital in Wuhan, China [2]. The clinical efficacy of raltegravir against neocrown pneumonia, the "hope of the people", will have to wait until the end of April to reveal the mystery.
Ridecivir was developed by Gilead Sciences, a company that has been working in the antiviral field (the anti-influenza drug oseltamivir, also known by its trade name Tamiflu, was first developed by Gilead and later sold to Roche for global distribution.) During the Ebola virus outbreak in 2013-2016 (West Africa) and 2018-2019 (Congo), major pharmaceutical companies around the world invested heavily in the development of anti-Ebola drugs. A preclinical pharmacodynamic study of raltegravir against Ebola, conducted by four of the industry's top laboratories - the U.S. Army Medical Research Institute of Infectious Diseases, Gilead Sciences, the CDC and Boston University School of Medicine - was published in the journal Nature in 2016 [3].
Molecular mechanism of raltegravir - Prodrug (Prodrug)
Triphosphate metabolite effectively stops RdRp enzyme activity
RNA-dependent RNA-polymerases (RdRp) are broad-spectrum targets for antiviral drug development, and most of the current anti-coronavirus drugs that target RdRp are nucleoside analogues or RNA interference classes [4]. After entering cells in the form of predrug (Prodrug), raltegravir is converted to the triphosphate metabolite NTP in three steps, and NTP and natural ATP compete to bind viral RdRp, which is inserted into the RNA synthesis chain, causing termination of viral RNA synthesis and inhibition of RdRp enzyme activity (Figure a below) [3].
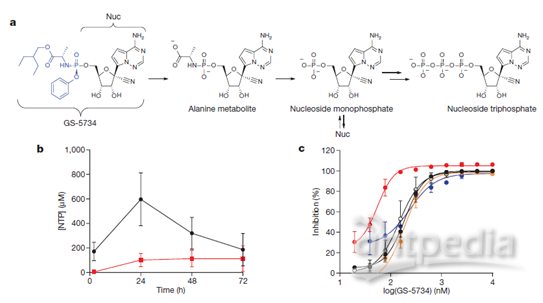
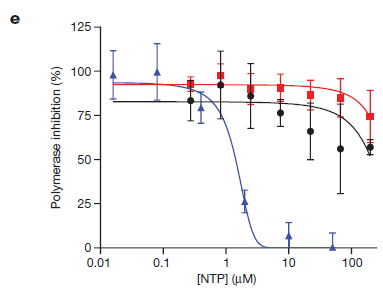
The 1'-cyano group on the structure of raltegravir, on the one hand, provides better binding activity against ATP competition for RdRpase and, on the other hand, provides better selective inhibition than human-derived RNA polymerase II and human-derived mitochondrial RNA polymerase (h-mtRNAP) against viral RdRpase. At the Hela cell level, raltegravir showed significant concentration-dependent inhibition against both Ebola viruses and three other viruses (lower panel c); and at the molecular level, the active molecule NTP of raltegravir selectively inhibited viral RdRpase activity (lower panel e blue), while no significant inhibition was observed against human-derived RNA polymerase II (lower panel e black) and mitochondrial RNA polymerase (lower panel e red) [3].
Ridcicevir cellular activity - highly efficient and selective
Inhibition of intracellular replication of the virus
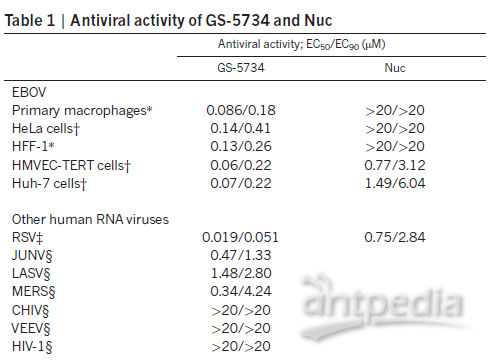
The researchers evaluated the antiviral activity of raltegravir (GS-5734) against Ebola virus and other RNA viruses on separate cellular models through further cytological assays. The data showed that raltegravir effectively inhibited Ebola activity on five cell models, including primary macrophages; and showed some inhibition of respiratory infection viruses, such as RSV and MERS, and hemorrhagic fever infection viruses, such as JUNV and LASV viruses; but no significant inhibition of other viruses, such as CHIV, VEEV and HIV-1, was observed (Table below) [3].
Ridciclovir in vivo efficacy - rapid diffusion to the lesion area, the
Improved survival rate of model animals
In rhesus monkeys, blood concentrations of raltegravir (black) and its metabolites, alanine metabolite (red), monophosphate metabolite Nuc (blue) and triphosphate metabolite NTP (green), were measured at different time points after intravenous administration at 10 mg/kg in healthy rhesus monkeys. The data show that the prodrug of raltegravir peaks in the body within two hours and is then rapidly cleared, while its triphosphate active metabolite NTP maintains high blood concentrations in the body, particularly in peripheral blood mononuclear cells (PBMCs), for a longer period of time. Further study of the drug in vivo division after labeling of raltegravir drug by isotope 14C revealed that the drug rapidly reaches the testes, epididymis, eyes and brain (Figure b below) [3].
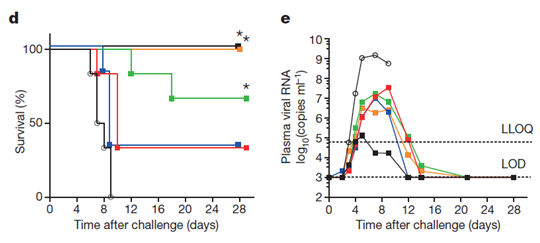
Through experiments in animal models of virus exposure, riddlesvir significantly increased the survival rate of rhesus monkey experimental animals after intravenous administration, especially in the group administered at a dose of 10 mg/kg after 3 days of virus exposure, whose survival rate after 28 days was as high as 100% as that of the blank control group (Figure d below), and it was further verified by nucleic acid quantification that the viral RNA copy number in the administered group was significantly suppressed compared to the blank control group (Figure e below) [3].
Summary of the antiviral drug mechanism of raltegravir
Ridecivir stands out among broad-spectrum antiviral nucleoside analogue inhibitors by using RdRpase as its drug target, mainly due to the following three points: 1) it has higher competitive affinity for its drug target, RdRpase, than its natural substrate, ATP; 2) at the in vitro cellular level, it can efficiently and selectively inhibit intracellular replication of RNA virus without significant cytotoxicity; 3) at the in vivo animal level, it has a good pharmacogenetic basis, its active metabolite NTP can rapidly diffuse to the lesion, inhibit RNA replication in vivo, and improve the survival rate of model animals after virus exposure.
[Disclaimer] This article comes from PerkinElmer Enterprise Management (Shanghai) Co., Ltd. The purpose of publishing this information on this site is to disseminate information and has nothing to do with the position of this site, nor does it constitute any investment or purchase advice for you.70 / 5,000