
![]()
Orforglipron: The "Disruptor" in the Oral GLP-1 Arena
While traditional GLP-1 drugs (such as Semaglutide, Tirzepatide) demonstrate significant efficacy, their injectable administration limits patient adherence. As a novel oral non-peptide small molecule drug, Orforglipron works through a dual mechanism: enhancing glucose-dependent insulin secretion and improving energy metabolism balance. It has shown glucose-lowering and weight-loss effects comparable to injectable formulations in Phase II clinical trials, with patients achieving up to a 13% reduction in body weight over a 26-week treatment period. The advantage of its oral dosage form expands the potential patient population by over 30%, marking the official entry of GLP-1 therapy into the "era of convenience."
Shandong Tianming Pharmaceutical: A Dual Commitment to Quality and Innovation
Shandong Tianming Pharmaceutical Group consistently upholds its mission of "Developing Innovative Medicines, Fulfilling Health Dreams," dedicated to providing global partners with customized intermediate solutions. Whether for rapid supply during early R&D stages or large-scale production for late-stage clinical trials, we will serve as your reliable partner with a rigorous approach, innovative technology, and stable quality.
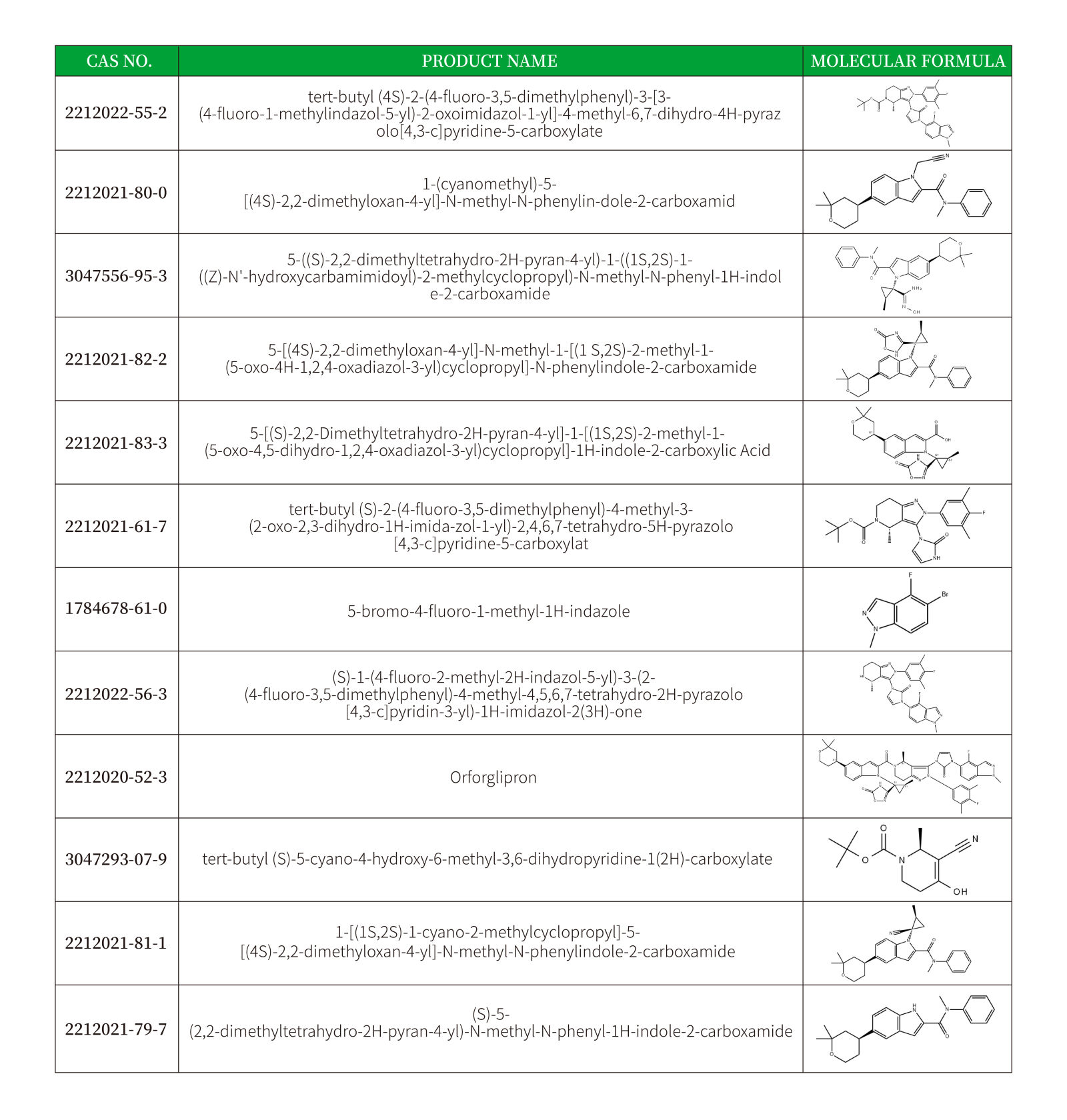
Jointly Reshaping the Global Landscape of Metabolic Disease Treatment
The formal commercialization of oral GLP-1 drugs is on the imminent horizon. According to Goldman Sachs predictions, the global market for metabolic disease drugs is projected to exceed $200 billion in the next decade, with oral dosage forms expected to capture over 40% of the market share. By providing cost-effective intermediates, Shandong Tianming Pharmaceutical empowers clients to accelerate new drug applications and jointly seize market opportunities.
Tianming Pharmaceutical Group offers stable supply of the Orforglipron intermediate series, from gram to kilogram scale. Inquiries are welcome for technical documentation and quotations on Orforglipron intermediates. Let's collaborate to advance the global adoption of oral GLP-1 therapies!